Womans Inoperable Brain Tumor Shrinks In Just Five Days Thanks To Cancer Breakthrough!
The landscape of modern oncology is frequently defined by incremental gains—months of added life, subtle shifts in survival percentages, or the stabilization of a chronic condition. However, every few decades, a clinical event occurs that fundamentally challenges our understanding of what is possible. In early 2024, researchers at Mass General Brigham initiated a phase 1 clinical trial that did exactly that. Targeting glioblastoma (GBM), a primary brain cancer notorious for its aggressive infiltration and resistance to conventional therapies, the team deployed a radical evolution of immunotherapy. The results were not just statistically significant; they were visually staggering. In one instance, a woman’s “inoperable” brain tumor began to regress in as little as forty-eight hours, signaling a potential paradigm shift in the fight against the most lethal form of brain cancer.
To understand the magnitude of this breakthrough, one must first appreciate the formidable nature of glioblastoma. GBM is often described by neuro-oncologists as a “multifaceted monster.” It does not grow as a single, solid mass with clear borders; instead, it sends out microscopic, thread-like tentacles that weave through healthy brain tissue. This makes complete surgical resection nearly impossible. Furthermore, the brain is protected by the blood-brain barrier (BBB), a highly selective semi-permeable border that prevents most standard chemotherapy drugs from reaching the tumor. For decades, the standard of care—a combination of surgery, radiation, and temozolomide—has offered only a modest extension of life, with recurrence being almost a statistical certainty.
The Mass General Brigham trial utilized an advanced form of CAR-T cell therapy, a treatment that has already revolutionized the management of liquid cancers like leukemia and lymphoma. CAR-T involves harvesting a patient’s own T-cells—the “soldiers” of the immune system—and genetically engineering them in a laboratory to express chimeric antigen receptors (CARs). These receptors act as a specialized GPS, allowing the T-cells to recognize and attach to specific proteins found on the surface of cancer cells. Once infused back into the patient, these “living drugs” hunt down and destroy the malignancy.
However, applying CAR-T to solid tumors in the brain has historically been met with failure. Glioblastomas are “heterogeneous,” meaning the cells within a single tumor are not all the same. If a CAR-T cell is programmed to target only one protein, the tumor cells that lack that protein will simply continue to grow, a phenomenon known as “antigen escape.” To bypass this defense, the Massachusetts researchers developed a dual-action strategy. They combined the CAR-T cells with “T-cell engaging antibody molecules” (TEAMs). This bi-specific approach essentially sharpened the immune system’s aim, allowing the engineered cells to recognize multiple markers simultaneously and recruit nearby, non-engineered immune cells to join the attack.
The delivery method was equally critical. Rather than infusing the cells intravenously, where they might be filtered out by the lungs or blocked by the blood-brain barrier, doctors delivered the treatment directly into the cerebrospinal fluid (CSF) via an Ommaya reservoir—a small device implanted under the scalp. This put the engineered immune cells face-to-face with the tumor in the fluid-filled spaces of the brain and spinal cord, bypassing the body’s natural checkpoints and maximizing the “kinetic energy” of the immune response.
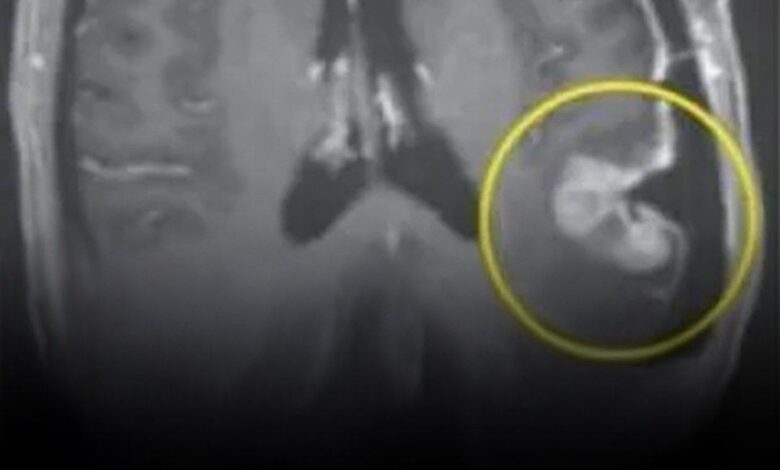
The clinical response was immediate and, in the words of the investigators, “stunned” the medical community. The trial involved three patients with recurrent glioblastoma—cases where all other treatments had failed. In the first patient, a 72-year-old man, the tumor shrank by 18.5% within just two days of a single infusion. By the tenth week, the tumor had regressed by over 60%. The second patient, a 57-year-old woman, saw her tumor nearly disappear on MRI scans within five days. The third patient, a 74-year-old man, showed similar rapid regression in a timeframe previously thought impossible for a biological therapy.
These results are described by experts as “early and fragile.” It is important to maintain a clinical perspective: this was a phase 1 trial with only three participants. While the initial regression was dramatic, the researchers are still monitoring for long-term durability and potential toxicity. The brain is an incredibly sensitive environment; a hyper-aggressive immune response can lead to neuro-inflammation or brain swelling, which must be carefully managed with steroids and other interventions. This is not yet a “cure” in the definitive sense, but it is an undeniable proof of concept. It proves that the immune system, when given the right tools and the right “address,” can penetrate the defenses of a glioblastoma and induce rapid cell death.
The implications for the future of neuro-oncology are profound. For a cancer long defined by a sense of clinical hopelessness, this trial marks the first time that a targeted immunotherapy has produced such a rapid, visible reduction in GBM mass. It opens the door for a new generation of “next-day” therapies where the timeframe for assessing treatment efficacy moves from months to days. Furthermore, the success of the TEAMs antibody integration suggests that the “antigen escape” problem—the primary hurdle for CAR-T in solid tumors—might finally have a technological solution.
As we move forward through 2026, larger trials are already being organized to determine if these results can be replicated across a broader patient population and if they can be translated into long-term remission. The goal is to move beyond the “one-off” success story and create a standardized protocol that can be deployed at cancer centers worldwide. For the thousands of families currently facing a glioblastoma diagnosis, the Mass General Brigham trial represents more than just a headline; it represents a glimpse of a future where a terminal diagnosis can be met with a viable, potent, and immediate counter-attack.
The story of the woman whose “inoperable” tumor shrank in five days is a testament to the power of precision medicine and the persistence of the human spirit. It serves as a reminder that even the most formidable biological monsters have a weakness. In the sterile, high-tech labs of Boston, the first cracks in the glioblastoma’s armor have finally appeared, and the light coming through those cracks is the brightest it has been in decades.